
This is just the heater power times the time. 2 to get the total internal energy of the container contents U.įor a constant volume system, you know from the first law of thermodynamics that $Q=\Delta U$, where Q is the total amount of heat you have added to get from the initial temperature to temperature T. 1 to solve for the mass fraction vapor x. $$m(1-x)v_L+mxv_V=V\tag$$where x is the mass fraction of water that is vapor. For any chosen temperature T, you look up in the saturated steam tables the specific volume of liquid water $v_L$, the specific volume of water vapor $v_V$, the specific internal energy of the liquid $u_L$, the specific internal energy of the vapor $u_V$, and, of course, the saturation pressure P. If you know the total volume of the container V and the total mass of water in the container m, then using the steam tables to get what you want is relatively straightforward. This seems like it must be a solved problem, but I'm struggling to find an approach to developing even an approximately solution to estimate how quickly pressure will increase over time. But it's not at all obvious to me how quickly it will increase in this dynamic system. Of course, the water is still evaporating at this sub-critical temperature, and heat energy is still being supplied at the rate of 1000W, so the pressure will continue to climb. My reasoning is that as the pressure builds, the water is suddenly below the new boiling point of the water at the increased pressure, and thus the rate of vaporization will slow as the temperature of the water lags behind the increasing boiling point. 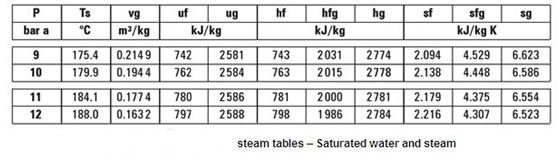
My naive assumption was that I could simply use the ideal gas law and solve for delta-P, but now I don't think that's correct. Initially, the boiling point of water at standard pressure is 100 ☌, and the latent heat of vaporization is 2257 Joules/gram, and I assume I'm adding 1000W = 1000Joules/sec, so I'm converting 0.443 grams/second into steam. I'm trying to think about how fast the rate of pressure will increase in an idealized boiler filled with water starting at 100☌, standard pressure, and supplied with 1000W of power.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
